
Kara L. Bren
she/her/hers
Richard S. Eisenberg Professor in Chemistry in the School of Arts & Sciences
Professor of Chemistry
Chair, Department of Chemistry
PhD, California Institute of Technology, 1996
- Office Location
- 448 Hutchison Hall
- Telephone
- (585) 275-4335
- Web Address
- Website
Research Overview
Professor Bren's research interests lie in the fields of bioinorganic and biophysical chemistry. The Bren group has developed new methods for engineering and characterizing metalloproteins in the cytochrome c family, contributing to our understanding of how protein structure dictates protein dynamics and function. In ongoing work, we are employing a range of spectroscopic techniques to understand how the three-dimensional structure of metalloproteins influences their electronic structure and reactivity.
In another major effort, the Bren group is developing metalloprotein- and metallopeptide-based catalysts with particular relevance to energy conversion. Metallobiomolecular catalysts offer a number of advantages including being water soluble and environmentally friendly. Furthermore, the use of protein engineering facilitates the preparation of multifunctional and modular structures and the manipulation of second-sphere interactions within the catalytic site.
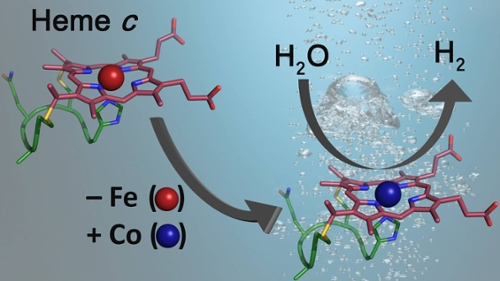
A current emphasis in the lab is the development of artificial hydrogenases, or enzymes that catalyze the reduction of protons derived from water to hydrogen (H2). Hydrogen produced from water is a green, carbon-free fuel, and hydrogen evolution driven by sunlight stores solar energy in the form of chemical bonds. The Bren group's artificial hydrogenases couple to synthetic, biomolecular, or nanotechnological photosensitizers to yield environmentally friendly assemblies to produce hydrogen as a solar fuel. The ultimate goal is to produce elegant and efficient systems that perform artificial photosynthesis.
Research Interests
- Bioinorganic and biophysical chemistry
Selected Publications
- Homogeneous Multielectron Electrocatalytic Reduction of Nitrate by an Iron Catalyst in Water. Kaye L. Kuphal, Jesse R. Stroka, Maria Fernandez Lizarazo, Shamitri Bandyopadhyay, William B. Brennessel, Kara L. Bren, JACS Au, 2025, 5, 3182-3188. DOI: 10.1021/jacsau.5c00361.
- Electrocatalytic CO2 Reduction by a Synthetic Cobalt Porphyrin Mini-Enzyme. Alison A. Salamatian, Jose L. Alvarez-Hernandez, Karishma B. Ramesh, Linda Leone, Angela Lombardi, Kara L. Bren, Chem. Sci., 2025, 16, 5707-5716.
- Engineered Metallobiocatalysts for Energy-Relevant Reactions, Ryan M. Kosko, Kaye Kuphal, Alison Salamatian, and Kara L. Bren, Curr. Opin. Chem. Biol., 2025, 84, 102545. DOI: 10.1016/j.cbpa.2024.102545
- Amphiphilic, Phosphonic Acid-Capped Cadmium Selenide Quantum Dots Sensitize a Thiomolybdate Catalyst for Hydrogen Production, Ryan Kosko, Qirui Wang, Chari Y.M. Peter, Todd D. Krauss, Kara L. Bren, and Ellen M. Matson, Chem. Comm., 2024, 60, 13694-13697. DOI: 10.1039/D4CC03656E
- Shewanella oneidensis MR-1 Respires CdSe Quantum Dots for Photocatalytic Hydrogen Evolution. Emily H. Edwards, Jana Jelušić, Ryan M. Kosko, Kevin P. McClelland, Soraya S. Ngarnim, Wesley Chiang, Sanela Lampa-Pastirk, Todd D. Krauss, Kara L. Bren. Proc. Natl. Acad. Sci. USA, 2023, 120, e2206975120. DOI: 10.1073/pnas.2206975120
- Potential- and Buffer-Dependent Selectivity for the Conversion of CO2 to CO by a Cobalt Porphyrin-Peptide Electrocatalyst in Water, Jose L. Alvarez-Hernandez,‡ Alison A. Salamatian,‡ Ji Won Han, and Kara L. Bren,(‡contributed equally), ACS Catal., 2022, 12, 14689-14697. DOI: 10.1021/acscatal.2c03297
- Linear Free Energy Relationships in Hydrogen Evolution Catalysis by a Cobalt Tripeptide in Water, Jose L. Alvarez-Hernandez, Jiwon Han, Andrew E. Sopchak, Yixing Guo, and Kara L. Bren, ACS Energy Lett., 2021, 6, 2256-2261. DOI: 10.1021/acsenergylett.1c00680
- Contributions to Cytochrome c Inner- and Outer-Sphere Reorganization Energy. Samir Chattopadhyay, Manjistha Mukherjee, Banu Kandemir, Sarah E. J. Bowman, Kara L. Bren, Abhishek Dey, Chem. Sci, 2021, 12, 11894–11913. DOI: 10.1039/D1SC02865K
- Electrocatalytic Multielectron Nitrite Reduction in Water by an Iron Complex, Jesse R. Stroka, Banu Kandemir, Ellen M. Matson, Kara L. Bren, ACS Catal., 2020, 10, 13968–13972. DOI: 10.1021/acscatal.0c03600
