Department News
Is a ‘quantum leap’ in chemistry in the offing?
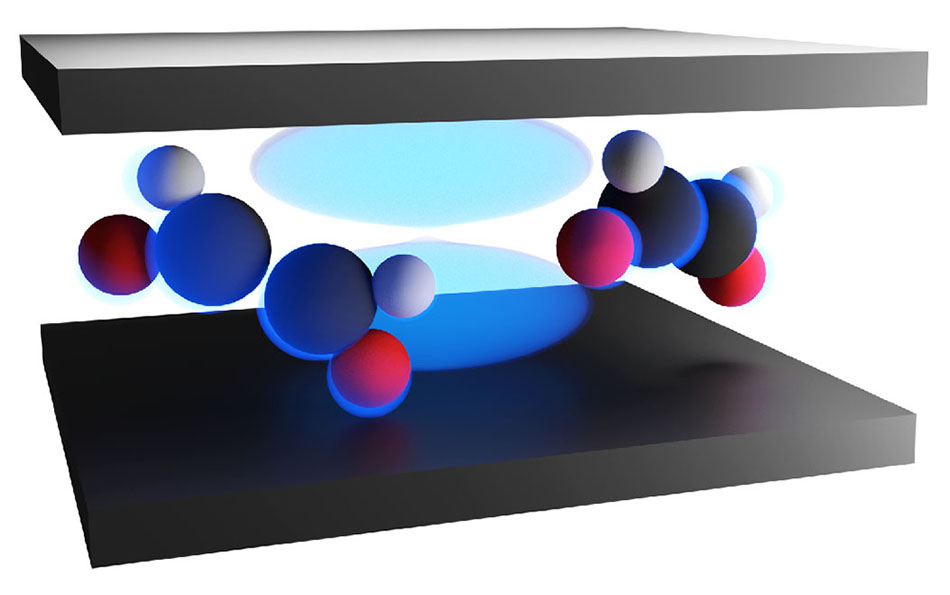
Artist’s rendering of an optical cavity containing two molecules. The Frank Huo lab at the University of Rochester has demonstrated how photons trapped in an optical cavity can control reactions between molecules. The light blue areas show the radiation fields in which photon-matter hybrid states, or polaritons, are created. (Courtesy of the Huo laboratory)
Researchers at the University of Rochester have come up with a theoretical framework demonstrating how photons trapped in optical cavities are able to control chemical reactions.
“I think this will facilitate a quantum leap in chemistry,” says Pengfei “Frank” Huo, assistant professor of chemistry. “It will likely inspire new principles in chemistry, taking advantage of the quantum mechanical nature of photons to enable new ‘tuning knobs’ that are usually not accessible to chemists.”
The findings, published in The Journal of Physical Chemistry Letters, help explain the results of experiments first conducted by the Thomas Ebbesen research group at the University of Strasbourg, France, seven years ago. Ebbesen’s group showed that the reactivity of molecules – and the energy transfer between them -- could be profoundly altered when the molecules were placed in optical cavities.
Optical cavities are devices, usually an arrangement of mirrors, in which photons – the fundamental particles of light – can be trapped. Physicists and optical engineers have traditionally used them to study the interaction of light with individual atoms and with solid state materials that have a very high degree of symmetry, Huo says.
Chemists, on the other hand, have used light primarily as an energy source to heat up or “excite” chemicals in order to conduct photochemical reactions.
“Nobody had really thought about putting a molecular system into quantum optic devices, and using trapped photons as chemical reagents, until the ground breaking Ebbesen experiments ” Arkajit Mandal says, who is lead author of the paper and a PhD student in the Huo group.
Further experiments have followed. However, “what has lagged behind is a rigorous theoretical understanding” of how this process actually occurs, Huo says.
The Huo group employed the University’s Blue Hive supercomputer to simulate the hybridized light/matter states (polaritons) that occur when molecules interact with photons in an optical cavity.
They demonstrated that, by “tuning the fundamental properties of a photon, such as its frequency or the photon-matter coupling strength,” the reactivity of molecules could also be manipulated in the cavity.
They also found that a photon could be recycled among many molecules to enable excited state reactions, “thus effectively functioning as a catalyst.”
“I think this is a great way to unify the two fields of photochemistry and quantum optics, and to enable the interplay of electronic, nuclear, and photonic degrees of freedom,” Huo says. “This rich interplay has already been shown to enable new chemical reactions, and no doubt many more we don’t realize yet.”
The work was supported with a National Science Foundation “Enabling Quantum Leap in Chemistry” grant. Other related experimental work in polariton chemistry at Rochester is taking place in Todd Krauss’ group in the Department of Chemistry and Nick Vamivakas’ group at the Institute of Optics. Huo is collaborating with both Krauss and Vamivakas to investigate polariton mediated charge transfer reactions.
Since starting his independent career at Rochester, Huo developed simulation methods to study electron and coupled proton-electron transfer in light-harvesting systems, which led to an NSF CAREER award. He found that the same methods could also be applied in this new work with hybridized electron/photon polariton states.
“That’s what got us very excited about this area in general,” Huo says, “where you can map one type of new problem onto the old problems you know how to solve, and just use the old tools to do it.”
Related Links...
- Author: Bob Marcotte, bmarcotte@ur.rochester.edu
- The Journal of Physical Chemistry Letters: Investigating New Reactivities Enabled by Polariton Photochemistry
- Huo Faculty Page
- Huo Research Group
