
Michael Welte
he/him/his
Professor
PhD
Research Active
Now accepting:
Undergraduate researchers
There may be openings for the spring and summer of 2026. Please e-mail with inquiries.
- Office Location
- 317 Hutchison
- Telephone
- (585) 276-3897
- Web Address
- Website
Office Hours: By appointment
Research Overview
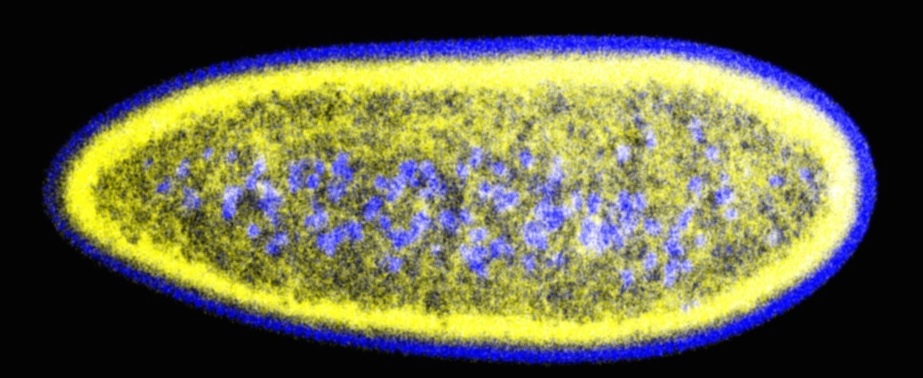
Lipid droplets (yellow) and nuclei (blue) in early Drosophila embryos
Cells are highly dynamic systems, in which many components are in constant flux: these include molecules, supramolecular complexes, and even entire organelles. They are continually generated, reorganized, relocated, and destroyed. Intracellular trafficking is highly directed in space and carefully controlled in time. The Welte laboratory studies the molecular mechanisms and biological roles of two types of trafficking events: the exchange of proteins between lipid droplets, cytoplasmic fat storage organelles, and the nucleus, as well as the motor-powered transport of various cargoes along microtubules. We employ Drosophila models in which we can combine in vivo visualization with molecular genetics approaches. We dissect the molecular mechanisms of these trafficking events and determine the consequences of disrupted trafficking at the organismal level. Our research is very visual and generates striking images (see the examples on the lab website). Currently, we work in three general areas: the role of lipid droplets as sequestration sites for nuclear proteins, the mechanism by which a nesprin family member modulates motor-driven transport, and the temporal regulation of lipid-droplet motion. For more information on our research, please check out the publications below. A full list of publications is available at our lab website.
Lipid droplets as protein sequestration sites
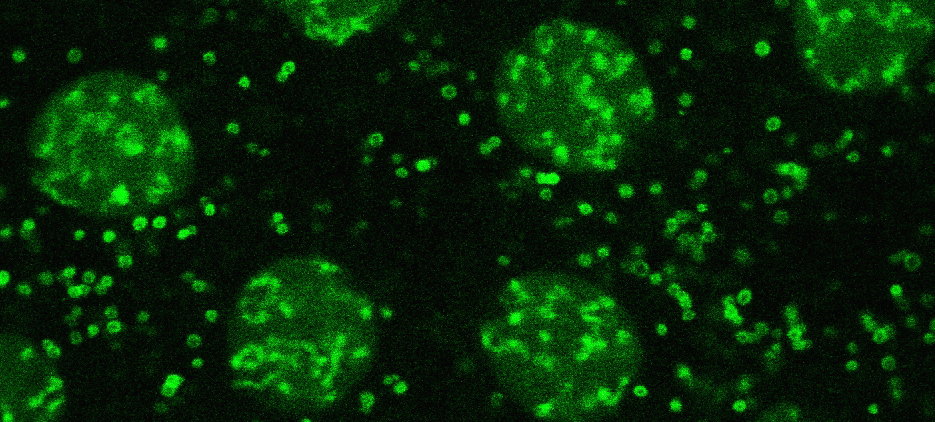
The variant histone H2Av (green) is present on lipid droplets and in nuclei
Lipid droplets are critical for lipid metabolism and energy homeostasis. We found that in Drosophila embryos lipid droplets also sequester massive amounts of certain histones (Cermelli et al., 2006), via the novel histone anchor Jabba (Li et al., 2012). Droplet-bound histones can be released and travel to the nuclei where they support chromatin assembly. In addition, droplet-bound histones mediate anti-bacterial defense (Anand et al., 2012). These studies revealed that in addition to their role in lipid metabolism lipid droplets also function as sites for regulated protein sequestration (Welte, 2007). For histones, such sequestration is important both for long-term storage (Li et al., 2012) and short-term buffering (Li et al., 2014). We are characterizing how Jabba recruits histones to lipid droplets, how the cell regulates histone release from droplets and their transfer to nuclei, and how droplet-bound histones contribute to innate immunity.
Motor regulation via the Klarsicht protein
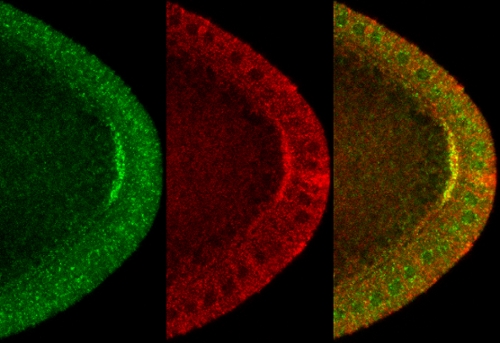
Colocalization of Klar (green) and Kinesin-1 (red) at the posterior pole of mid-stage oocytes
The nesprin family of proteins plays critical roles in linking the cytoskeleton to the nucleoskeleton. This protein family also acts in the cytoplasm, but these functions remain poorly understood. We discovered that the Drosophila family member Klarsicht (Klar) exists in complexes with microtubule motors and regulates motor-driven transport of lipid droplets in early embryos (Welte et al., 1998; Guo et al., 2005; Yu et al., 2011) and of oskar mRNPs during oogenesis (Gaspar et al., 2014). Depending on context, Klar acts either as positive or negative regulator of kinesin-1 and may be an integral component of a still mysterious switching mechanism that turns motor activity on or off. This fine-tuning ability allows Klar to compensate for temperature-induced imbalance between RNP motility and RNP anchoring, thus contributing to developmental robustness (Gaspar et al., 2014). We are now determining how Klar interacts with microtubule motors and how it adapts transport to temperature.
Temporal regulation of lipid-droplet motion
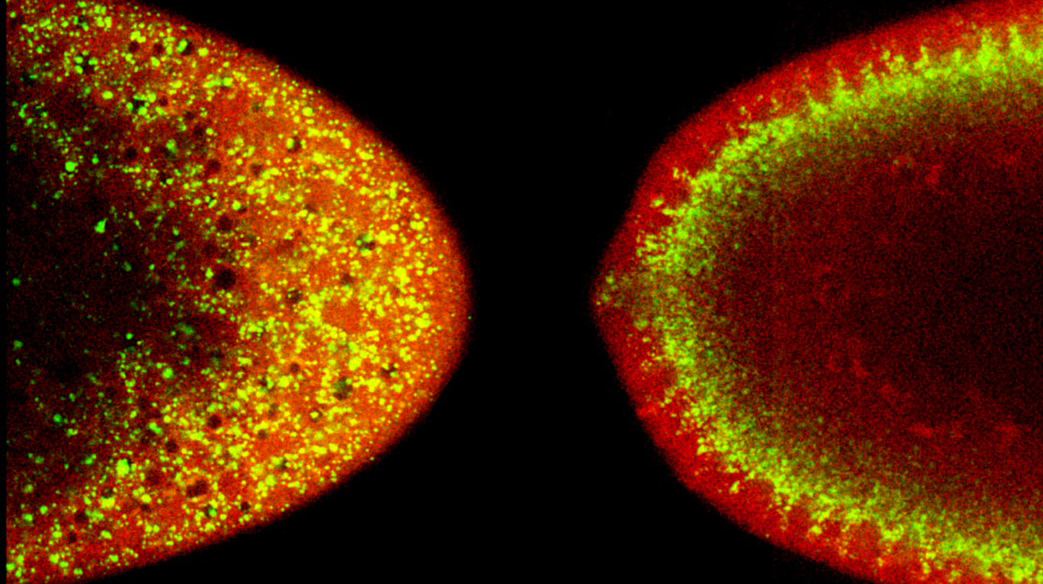
Lipid droplets (yellow) undergo global shifts during embryogenesis
Lipid droplets are motile in many cells, including in early Drosophila embryos (Welte, 2009). Here, droplets move bidirectionally along microtubules, powered by the motors kinesin-1 and cytoplasmic dynein (Shubeita et al. 2008; Gross et al., 2000). As embryogenesis proceeds, the balance of kinesin- and dynein-based motion is intricately regulated, causing redistribution of the droplet population as a whole. Temporal regulation is achieved via the Halo protein (Gross et al., 2003), which - directly or indirectly – controls the properties of kinesin-1 on droplets. We are determining the molecular mechanism by which Halo alters kinesin-1 function as well as the pathways that control Halo expression.
Research Interests
- Cell and developmental biology
- Intracellular trafficking of proteins, RNAs, and organelles
- Regulation of motor-driven transport
- Cell biology of lipid droplets
- Lipid droplets in protein sequestration and innate immunity
Recent Publications
A role for triglyceride lipase brummer in the regulation of sex differences in Drosophila fat storage and breakdown PLOS Biology, 2020 https://doi.org/10.1371/journal.pbio.3000595
Lipid Droplet Motility and Organelle Contacts Contact, 2019 https://doi.org/10.1177/2515256419895688
Francesca Nardi, Philip Fitchev, Kyrsten M. Brooks, Omar E. Franco, Kevin Cheng, et al. Lipid droplet velocity is a microenvironmental sensor of aggressive tumors regulated by V-ATPase and PEDF Laboratory Investigation, 2019
Fanning S, Haque A, Imberdis T, Baru V, Barrasa MI, et al. Lipidomic Analysis of α-Synuclein Neurotoxicity Identifies Stearoyl CoA Desaturase as a Target for Parkinson Treatment. Molecular cell, 2019 https://doi.org/10.1016/j.molcel.2018.11.028
Johnson MR, Stephenson RA, Ghaemmaghami S, Welte MA Developmentally regulated H2Av buffering via dynamic sequestration to lipid droplets in Drosophila embryos. eLife, 2018 https://doi.org/10.7554/elife.36021
Nardi F, Fitchev P, Franco OE, Ivanisevic J, Scheibler A, et al. PEDF regulates plasticity of a novel lipid-MTOC axis in prostate cancer-associated fibroblasts. Journal of cell science, 2018 https://doi.org/10.1242/jcs.213579
Pennetta G, Welte MA Emerging Links between Lipid Droplets and Motor Neuron Diseases. Developmental cell, 2018 https://doi.org/10.1016/j.devcel.2018.05.002
, Lipid droplet functions beyond energy storage. Biochimica et biophysica acta, 2017 https://doi.org/10.1016/j.bbalip.2017.07.006
, , , , , et al. A Luciferase-fragment Complementation Assay to Detect Lipid Droplet-associated Protein-Protein Interactions. Molecular & cellular proteomics : MCP, 2017 https://doi.org/10.1074/mcp.m116.061499
, , , , Temporal control of bidirectional lipid-droplet motion in Drosophila depends on the ratio of kinesin-1 and its co-factor Halo. Journal of cell science, 2016 https://doi.org/10.1242/jcs.183426
For more, please visit my ORCID Profile
