
Andrei Seluanov
Dean’s Professor
Professor of Biology
PhD
Research Active
Now accepting:
PhD students
Please email with inquiries.
Undergraduate researchers
Please email with inquiries.
- Office Location
- 432 Hutchison
- Telephone
- (585) 275-6636
- Web Address
- Website
Office Hours: By appointment
Research Overview
Our research focus is on understanding the mechanisms of longevity and cancer resistance. Aging is the major cause of death in developed countries. By finding ways to delay aging it will be possible to delay the onset of multiple age-related diseases. Cancer is another major killer in developed world, where 25% of human mortality is caused by cancer. Cancer incidence increases exponentially with age and to achieve long-life species must evolve efficient tumor suppressor mechanisms. Our goal is to understand such mechanisms in mammalian species that are naturally cancer-resistant.
Anticancer mechanisms in the long-lived rodents the naked mole-rat and Eastern grey squirrel
Cancer affects most, if not all, classes of vertebrates. The major tumor suppressor pathway appear to be conserved among mammalian species, however, the cancer rates are strikingly different. For example, up to 95% of mice die from cancer, whereas another rodent, the naked mole rat appears to be cancer proof. Human cancer mortality is 25%, which is in somewhat in-between the mouse and the naked mole-rat. In addition to its cancer resistance naked mole-rat is highly interesting due to its longevity. It has the maximum lifespan of 30 years, which is almost 10 times longer than a similar size mouse. We recently discovered that the naked mole-rat has a novel anticancer mechanism named “early contact inhibition” that contribute to its cancer resistance. We are interested in the genes and signaling pathways involved in early contact inhibition and in other mechanisms that contribute to naked mole-rat longevity and cancer resistance. Eastern grey squirrel is another very interesting rodent. Grey squirrels are common in cities and parks across the United States, but a few people know that these rodents have a maximum lifespan of 24 years. Squirrels express extremely high telomerase activity in all tissues, comparable to human tumor tissue. High telomerase activity is generally associated with increased cancer risk, therefore squirrels must possess unique mechanisms that allow them to stay cancer free despite the high telomerase activity.

Sirtuins
Yeast Sir2 gene is a histone deacetylase involved in gene silencing. Furthermore overespression of Sir2 extends yeast lifespan and promotes genome stability. Mammals have seven homologs of Sir2 named sirtuins (SIRT1-SIRT7). Deletion of mouse SIRT6 leads to premature aging and genomic instability. We are interested in the role of SIRT6 in genome stability and stress resistance.
Anticancer mechanisms in whales
If all mammals were equally susceptible to oncogenic mutations and had equal tumor suppressor mechanisms one would expect that the rate of cancer would be proportional to the species body size and longevity. This is because greater number of cells and greater number of cell divisions would increase the chances of malignant transformation. Mammals range in body size from 30 g mouse to 110,000,000 g blue whale. If up to 95% of mice die of cancer by the age of 2 years it is paradoxical how come blue whales do not succumb to cancer in the womb. This fundamental question is named “Peto’s paradox” after Sir Richard Peto, who described it in 1975. The most plausible solution to Peto’s paradox is that the large and long-lived species have evolved superior anticancer systems. We are interested in understanding these mechanisms with the goal of using them to prevent human cancer.
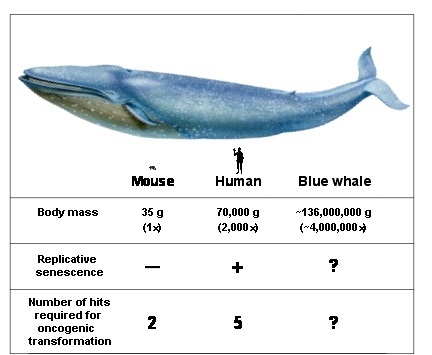
Enumeration of anticancer mechanisms in mouse, human, and blue whale. These three species differ in body mass by a factor of 2,000: human is 2,000 times larger than a mouse, and blue whale is 2,000 times larger than a human. Humans have multiple additional anticancer adaptations compared to mice. It is currently unknown whether whales have evolved many additional anticancer adaptations compared to humans. Body mass in large whales is difficult to measure. Therefore, the figures are approximate estimates.
To learn more about our research, please visit our lab website Gorbunova Lab.
Research Interests
- Molecular and cell biology
- Tumor suppressor mechanisms in log-lived rodents the Naked Mole-Rat and Eastern Grey Squirrel
- Mammalian Sirt proteins in regulation of stress response during aging
- Anti-cancer mechanisms in whales
Selected Publications
Peer Reviewed Publications
- Quanwei Zhang, Gregory Tombline, Julia Ablaeva, Lei Zhang, Xuming Zhou, Smith Zachary, Alus M. Xiaoli, Zhen Wang, Jhih-Rong Lin, M. Reza Jabalameli, Joydeep Mitra, Nha Nguyen, Jan Vijg, Andrei Seluanov, Vadim N. Gladyshev, Vera Gorbunova, Zhengdong D. Zhang. (2021) North American beaver genome provides insights into the mechanisms of longevity and cancer resistance. (Cell Reports) In revision.
- Stephan Emmrich, Marco Mariotti, Masaki Takasugi, Maggie E. Straight, Alexandre Trapp, Vadim N. Gladyshev, Andrei Seluanov, Vera Gorbunova (2021) The hematopoietic landscape at single-cell resolution reveals unexpected stem cell features in naked mole-rats. (Cell) In revision.
- Matthew Simon, Jiping Yang, Jon Gigas, Tory M. Schaff, Lei Zhang, Maria Zagorulya, Greg Tombline, Michael Gilbert, Samantha L. Yu, Alexis Pope, Michael Van Meter, Stephan Emmrich, Jeehae Han, Seungjin Ryu, Archana Tare, Yizhou Zhu, Adam Hudgins, Gil Atzmon, Nir Barzilai, Aaron Wolfe, Kelsey Moody, Benjamin A. Garcia, David D. Thomas, Paul D. Robbins, Jan Vijg, Andrei Seluanov, Yousin Suh, Vera Gorbunova (2021) A rare variant of SIRT6 identified in human centenarians improves genome maintenance and shows stronger interaction with Lamin A. (Nature) Under review.
- Yang Zhao, Ena Oreskovic, Quanwei Zhang, Quan Lu, Abbey Gilman, Junyue He, Jina Lee, Zhonghe Ke, Julia Ablaeva, Matthew Sweet, Steve Horvath, Zhengdong Zhang, Eviatar Nevo, Andrei Seluanov, Vera Gorbunova (2021) Transposon-triggered innate immune response confers cancer resistance to the blind mole rat. (Nature) Under review.
- Vera Gorbunova, Andrei Seluanov, Paolo Mita, Jef D. Boeke, Sara B. Linker, Fred H. Gage, Jill A. Kreiling, Anna P. Petrashen, Trenton Woodham, Jackson R. Taylor, Stephen L. Helfand, John M. Sedivy. (2021) The role of retrotransposable elements in aging and age-associated diseases. Nature, Accepted.
- Pawelec G, Bronikowski A, Cunnane SC, Ferrucci L, Franceschi C, Fülöp T, Gaudreau P, Gladyshev VN, Gonos ES, Gorbunova V, Kennedy BK, Larbi A, Lemaître JF, Liu GH, Maier AB, Morais JA, Nóbrega OT, Moskalev A, Rikkert MO, Seluanov A, Senior AM, Ukraintseva S, Vanhaelen Q, Witkowski J, Cohen AA. (2020) The conundrum of human immune system "senescence". Mech Ageing Dev. 2020 Dec;192:111357.
- Taguchi T, Kotelsky A, Takasugi M, Chang M, Ke Z, Betancourt M, Buckley MR, Zuscik M, Seluanov A, Gorbunova V. (2020) Naked mole-rats are extremely resistant to post-traumatic osteoarthritis. Aging Cell. Nov;19(11):e13255.
- Ke Z, Firsanov D, Spencer B, Seluanov A, Gorbunova V. (2020) Short-term calorie restriction enhances DNA repair by non-homologous end joining in mice. NPJ Aging Mech Dis. Aug 14;6:9.
- Swovick K, Firsanov D, Welle KA, Hryhorenko J, Wise JP, George C, Sformo TL, Seluanov A, Gorbunova V, Ghaemmaghami S. (2020) Interspecies differences in proteome turnover kinetics are correlated with lifespans and energetic demands. Mol Cell Proteomics. Dec 28:mcp.RA120.002301.
- Simon M., Emmrich S., Seluanov A., Gorbunova V. (2020) A hairy tale: SIRT7 safeguards skin stem cells during aging. EMBO J. Sep 15;39(18):e106294.
- Kumar V, Agrawal R, Pandey A, Kopf S, Hoeffgen M, Kaymak S, Bandapalli OR, Gorbunova V, Seluanov A, Mall MA, Herzig S, Nawroth PP. (2020) Compromised DNA repair is responsible for diabetes-associated fibrosis. EMBO J. Jun 2;39(11):e103477.
- Gorbunova V., Masaki Takasugi M., Seluanov A. (2020) Hyaluronan goes to great length. Cell Stress, Jul 17;4(9):227-229.
- Takasugi M, Firsanov D, Tombline G, Ning H, Ablaeva J, Seluanov A, Gorbunova V. (2020) Naked mole-rat very-high-molecular-mass hyaluronan exhibits superior cytoprotective properties. Nat Commun. May 12;11(1):2376.
- Rezazadeh S, Yang D, Biashad SA, Firsanov D, Takasugi M, Gilbert M, Tombline G, Bhanu NV, Garcia BA, Seluanov A, Gorbunova V. (2020) SIRT6 mono-ADP ribosylates KDM2A to locally increase H3K36me2 at DNA damage sites to inhibit transcription and promote repair. Aging (Albany NY). Jun 25;12(12):11165-11184.
- Zhao J, Tian X, Zhu Y, Zhang Z, Rydkina E, Yuan Y, Zhang H, Roy B, Cornwell A, Nevo E, Shang X, Huang R, Kristiansen K, Seluanov A, Fang X, Gorbunova V. (2020) Reply to: Transformation of naked mole-rat cells. Jul;583(7814):E8-E13.
- Gorbunova V, Seluanov A, Kennedy BK. (2020) The World Goes Bats: Living Longer and Tolerating Viruses. Cell Metab. Jul 7;32(1):31-43.
- Zhou X, Dou Q, Fan G, Zhang Q, Sanderford M, Kaya A, Johnson J, Karlsson EK, Tian X, Mikhalchenko A, Kumar S, Seluanov A, Zhang ZD, Gorbunova V, Liu X, Gladyshev VN. (2020) Beaver and Naked Mole Rat Genomes Reveal Common Paths to Longevity. Cell Rep. Jul 28;32(4):107949.
- Tombline G, Gigas J, Macoretta N, Zacher M, Emmrich S, Zhao Y, Seluanov A, Gorbunova V. (2019) Proteomics of long-lived mammals. Nov 18:e1800416. doi: 10.1002/pmic.201800416.
- Katagi H, Louis N, Unruh D, Sasaki T, He X, Zhang A, Ma Q, Piunti A, Shimazu Y, Lamano JB, Carcaboso AM, Tian X, Seluanov A, Gorbunova V, Laurie KL, Kondo A, Wadhwani NR, Lulla R, Goldman S, Venneti S, Becher OJ, Zou L, Shilatifard A, Hashizume R. (2019) Radiosensitization by Histone H3 Demethylase Inhibition in Diffuse Intrinsic Pontine Glioma. Clin Cancer Res. 25(18):5572-5583.
- Chen Y, Zhang H, Xu Z, Tang H, Geng A, Cai B, Su T, Shi J, Jiang C, Tian X, Seluanov A, Huang J, Wan X, Jiang Y, Gorbunova V, Mao Z. (2019) A PARP1-BRG1-SIRT1 axis promotes HR repair by reducing nucleosome density at DNA damage sites. Nucleic Acids Res. 47(16):8563-8580.
- Rezazadeh S, Yang D, Tombline G, Simon M, Regan SP, Seluanov A, Gorbunova V. (2019) SIRT6 promotes transcription of a subset of NRF2 targets by mono-ADP-ribosylating BAF170. Nucleic Acids Res. 47(15):7914-7928.
- Simon M, Van Meter M, Ablaeva J, Ke Z, Gonzalez RS, Taguchi T, De Cecco M, Leonova KI, Kogan V, Helfand SL, Neretti N, Roichman A, Cohen HY, Meer MV, Gladyshev VN, Antoch MP, Gudkov AV, Sedivy JM, Seluanov A, Gorbunova V (2019) LINE1 derepression in aged wild type and SIRT6 deficient mice drives inflammation. Cell Metabolism, 29(4):871-885.
- De Cecco M, Ito T, Petrashen AP, Elias AE, Skvir NJ, Criscione SW, Caligiana A, Brocculi G, Adney EM, Boeke JD, Le O, Beauséjour C, Ambati J, Ambati K, Simon M, Seluanov A, Gorbunova V, Slagboom PE, Helfand SL, Neretti N, Sedivy JM. (2019) L1 drives IFN in senescent cells and promotes age-associated inflammation. Nature, 566(7742):73-78.
- Tian X, Firsanov D, Luo L, Tombline G, Henderson S, Steffan J, Tam J, Zheng K, Cornwell A, Johnson A, Yang J-N, Mao Z, Manta B, Gladyshev VN, Zhang Z, Vijg J, Seluanov A, Gorbunova V, (2019) SIRT6 is Responsible for More Efficient DNA Double-Strand Break Repair in Long-Lived Species. Cell, 177(3):622-638.
- Bourque G, Burns KH, Gehring M, Gorbunova V, Seluanov A, Hammell M, Imbeault M, Izsvák Z, Levin HL, Macfarlan TS, Mager DL, Feschotte C (2018) Ten things you should know about transposable elements. Genome Biol. 2018 19(1):199.
- Seluanov A, Gladyshev VN, Vijg J, Gorbunova V. (2018) Mechanisms of cancer resistance in long-lived mammals. Nature Reviews Cancer. 18(7):433-441.
- Ke Z,Seluanov A, Gorbunova V. (2018) Accurate translation is important for longevity. Aging (Albany NY). 10(3):297-298.
- Evdokimov A, Kutuzov M, Petruseva I, Lukjanchikova N, Kashina E, Kolova E, Zemerova T, Romanenko S, Perelman P, Prokopov D, Seluanov A, Gorbunova V, Graphodatsky A, Trifonov V, Khodyreva S, Lavrik O. (2018) Naked mole rat cells display more efficient excision repair than mouse cells. Aging (Albany NY). 10(6):1454-1473.
- Zhao Y, Tyshkovskiy A, Muñoz-Espín D, Tian X, Serrano M, de Magalhaes JP, Nevo E, Gladyshev VN, Seluanov A, Gorbunova V. (2018) Naked mole rats can undergo developmental, oncogene-induced and DNA damage-induced cellular senescence. Proc Natl Acad Sci U S A. 115(8):1801-1806.
- Swovick K, Welle KA, Hryhorenko J, Seluanov A, Gorbunova V, Ghaemmaghami S. (2018) Cross-species comparison of proteome turnover kinetics. Mol Cell Proteomics. 17(4):580-591.
- Tian X, Doerig K, Park R, Can Ran Qin A, Hwang C, Neary A, Gilbert M, Seluanov A, Gorbunova V. (2018) Evolution of telomere maintenance and tumour suppressor mechanisms across mammals. Philos Trans R Soc Lond B Biol Sci. 373; 20160443.
- Piscitello D, Varshney D, Lilla S, Vizioli MG, Reid C, Gorbunova V, Seluanov A, Gillespie DA, Adams PD. (2018) AKT overactivation can suppress DNA repair via p70S6 kinase-dependent downregulation of MRE11. Oncogene. 37(4):427-438.
- Tan L, Ke Z, Tombline G, Macoretta N, Hayes K, Tian X, Lv R, Ablaeva J, Gilbert M, Bhanu NV, Yuan ZF, Garcia BA, Shi YG, Shi Y, Seluanov A, Gorbunova V. (2017) Naked Mole Rat Cells Have a Stable Epigenome that Resists iPSC Reprogramming. Stem Cell Reports. 9(5):1721-1734.
- Tian X, Seluanov A, Gorbunova V. (2017) Molecular Mechanisms Determining Lifespan in Short- and Long-Lived Species. Trends Endocrinol Metab. 28(10):722-734.
- Ke Z, Mallik P, Johnson AB, Luna F, Nevo E, Zhang ZD, Gladyshev VN, Seluanov A, Gorbunova V. (2017) Translation fidelity coevolves with longevity. Aging Cell. 16(5):988-993.
- Tian X, Seluanov A, Gorbunova V. (2017) Beyond Making Ends Meet: DNA-PK, Metabolism, and Aging. Cell Metab. 25(5):991-992.
- Valenzano DR, Aboobaker A, Seluanov A, Gorbunova V. (2017) Non-canonical aging model systems and why we need them. EMBO J. 36(8):959-963.
- Moskalev A, Anisimov V, Aliper A, Artemov A, Asadullah K, Belsky D, Baranova A, de Grey A, Dixit VD, Debonneuil E, Dobrovolskaya E, Fedichev P, Fedintsev A, Fraifeld V, Franceschi C, Freer R, Fülöp T, Feige J, Gems D, Gladyshev V, Gorbunova V, Irincheeva I, Jager S, Jazwinski SM, Kaeberlein M, Kennedy B, Khaltourina D, Kovalchuk I, Kovalchuk O, Kozin S, Kulminski A, Lashmanova E, Lezhnina K, Liu GH, Longo V, Mamoshina P, Maslov A, Pedro de Magalhaes J, Mitchell J, Mitnitski A, Nikolsky Y, Ozerov I, Pasyukova E, Peregudova D, Popov V, Proshkina E, Putin E, Rogaev E, Rogina B, Schastnaya J, Seluanov A, Shaposhnikov M, Simm A, Skulachev V, Skulachev M, Solovev I, Spindler S, Stefanova N, Suh Y, Swick A, Tower J, Gudkov AV, Vijg J, Voronkov A, West M, Wagner W, Yashin A, Zemskaya N, Zhumadilov Z, Zhavoronkov A. (2017) A review of the biomedical innovations for healthy longevity. Aging (Albany NY). 9(1):7-25.
- Bozek K, Khrameeva EE, Reznick J, Omerbašić D, Bennett NC, Lewin GR, Azpurua J, Gorbunova V, Seluanov A, Regnard P, Wanert F, Marchal J, Pifferi F, Aujard F, Liu Z, Shi P, Pääbo S, Schroeder F, Willmitzer L, Giavalisco P, Khaitovich P. (2017) Lipidome determinants of maximal lifespan in mammals. Sci Rep. 7(1):5.
- Ma S, Upneja A, Galecki A, Tsai YM, Burant CF, Raskind S, Zhang Q, Zhang ZD, Seluanov A, Gorbunova V, Clish CB, Miller RA, Gladyshev VN. (2016) Cell culture-based profiling across mammals reveals DNA repair and metabolism as determinants of species longevity. eLife 5:e19130.
- Van Meter M, Simon M, Tombline G, May A, Morello TD, Hubbard BP, Bredbenner K, Park R, Sinclair DA, Bohr VA, Gorbunova V, Seluanov A. (2016) JNK Phosphorylates SIRT6 to Stimulate DNA Double-Strand Break Repair in Response to Oxidative Stress by Recruiting PARP1 to DNA Breaks. Cell Rep. 16(10):2641-50.
- Dokukin M, Ablaeva Y, Kalaparthi V, Seluanov A, Gorbunova V, Sokolov I. (2016) Pericellular Brush and Mechanics of Guinea Pig Fibroblast Cells Studied with AFM. Biophys J. 111(1):236-46.
- Hewitt G, Carroll B, Sarallah R, Correia-Melo C, Ogrodnik M, Nelson G, Otten EG, Manni D, Antrobus R, Morgan BA, von Zglinicki T, Jurk D, Seluanov A, Gorbunova V, Johansen T, Passos JF, Korolchuk VI. (2016) SQSTM1/p62 mediates crosstalk between autophagy and the UPS in DNA repair. 12(10):1917-30.
- Gorbunova V, Rezazadeh S, Seluanov A. (2016) Dangerous Entrapment for NRF2. Cell, 165(6):1312-3.
- Patrick A, Seluanov M, Hwang C, Tam J, Khan T, Morgenstern A, Wiener L, Vazquez JM, Zafar H, Wen R, Muratkalyeva M, Doerig K, Zagorulya M, Cole L, Catalano S, Lobo Ladd AA, Coppi AA, Coşkun Y, Tian X, Ablaeva J, Nevo E, Gladyshev VN, Zhang ZD, Vijg J, Seluanov A, Gorbunova V. (2016) Sensitivity of primary fibroblasts in culture to atmospheric oxygen does not correlate with species lifespan. Aging (Albany NY). 8(5):841-7.
- Gorbunova V, and Seluanov A. (2016) DNA double strand break repair, aging and the chromatin connection. Mutation Research, 788:2-6.
- Ma S, Lee SG, Kim EB, Park TJ, Seluanov A, Gorbunova V, Buffenstein R, Seravalli J, Gladyshev VN. 2015. Organization of the Mammalian Ionome According to Organ Origin, Lineage Specialization, and Longevity.Cell Reports13(7): 1319-26.
- Seluanov A, and Gorbunova V. 2015. The next top models: The naked truth. Cell163(1): 18.
- MacRae SL, Croken MM, Calder RB, Aliper A, Milholland B, White RR, Zhavoronkov A, Gladyshev VN, Seluanov A, Gorbunova V, Zhang ZD, and Vijg J. 2015. DNA repair in species with extreme lifespan differences.Aging (Albany NY). 2015 Dec; 7(12): 1171-84.
- Van Meter M, Seluanov A, and Gorbunova V. 2015. Wrangling Retrotransposons. The Scientist. March 1.
- MacRae SL, Zhang Q, Lemetre C, Seim I, Calder RB, Hoeijmakers J, Suh Y, Gladyshev VN, Seluanov A, Gorbunova V, Vijg J, and Zhang ZD. 2015. Comparative analysis of genome maintenance genes in naked mole rat, mouse, and human. Aging Cell. 14(2): 288-91.
- Tian X, Azpurua J, Gorbunova V, and Seluanov A. 2015. INK4 locus of the tumor-resistant rodent, the naked mole rat, expresses a functional p15/p16 hybrid isoform. Proc. Natl. Acad. Sci. USA, 112(4): 1053-8.
- Xu Z, Zhang L, Zhang W, Meng D, Zhang H, Jiang Y, Xu X, Van Meter M, Seluanov A, Gorbunova V, Mao Z. 2015. SIRT6 rescues the age related decline in base excision repair in a PARP1-dependent manner. Cell Cycle, 14(2): 269-76.
- VanMeter M, Gorbunova V, and Seluanov A. 2014. SIRT6: a promising target for cancer prevention and therapy, in “Anticancer Genes”, Editor: Grimm S., Springer.
- Keane M, Craig T, Alföldi J, Berlin AM, Johnson J, Seluanov A, Gorbunova V, Di Palma F, Lindblad-Toh K, Church GM, de Magalhães JP. 2014. The Naked Mole Rat Genome Resource: facilitating analyses of cancer and longevity-related adaptations. Bioinformatics.30(24): 3558-60.
- Van Meter M, Kashyap M, Sarallah R, Tian X, Seluanov A, and Gorbunova V. 2014. SIRT6 represses LINE1s by ribosylating Kap1 and the repression fails with aging and stress. Nature Communications, 5: 5011.
- Vaidya A, Mao Z, Tian X, Spencer B, Seluanov A, Gorbunova V. 2014. Knock-in reporter mice demonstrate that DNA repair by non-homologous end joining declines with age. PLoS Genetics10(7): e1004511.
- Fang X, Nevo E, Han L, Levanon EY, Zhao J, Avivi A, Larkin D, Jiang X, Feranchuk S, Zhu Y, Fishman A, Feng Y, Sher N, Xiong Z, Hankeln T, Huang Z, Gorbunova V, Zhang L, Zhao W, Wildman DE, Xiong Y, Gudkov A, Zheng Q, Rechavi G, Liu S, Bazak L, Chen J, Knisbacher BA, Lu Y, Shams I, Gajda K, Farré M, Kim J, Lewin HA, Ma J, Band M, Bicker A, Kranz A, Mattheus T, Schmidt H, Seluanov A, Azpurua J, McGowen MR, Ben Jacob E, Li K, Peng S, Zhu X, Liao X, Li S, Krogh A, Zhou X, Brodsky L, Wang J. 2014. Genome-wide adaptive complexes to underground stresses in blind mole rats Spalax. Nature Communications, 5: 3966.
- Gorbunova V, Seluanov A, Zhang Z, Gladyshev V, and Vijg J. 2014. Comparative genetics of longevity and cancer: insights from long-lived rodents. Nature Reviews Genetics, 15(8): 531-40 (Invited review).
- Hine CM, Li H, Xie L, Mao Z, Seluanov A, Gorbunova V. 2014. Regulation of Rad51 promoter. Cell Cycle13(13): 2038-45.
- XuY, CaoY, LiZ, JiangY, SeluanovA, GorbunovaV, and Mao Z. 2014. Utilization of Rad51C promoter for selective killing of cancer cells. Oncotarget,5(7): 1805-11.
- KeZ., VaidyaA., Jill Ascher J., Seluanov A. and Gorbunova V. 2014. Novel husbandry techniques support survival of naked mole-rat (Heterocephalus glaber) pups. J Am Assoc Lab Anim Sci., 53(1): 89-91.
- AzpuruaJ., KeZ., Chen I., ZhangQ., ErmolenkoD.N., Zhendong D. Zhang Z.D., Gorbunova V. SeluanovA. 2013. Naked mole-rat has increased translational fidelity compared with the mouse, as well as a unique 28S ribosomal RNA cleavage.Proc. Natl. Acad. Sci. USA, 110(43): 17350-5.
- Yang J-N, Seluanov A., Gorbunova V. 2013. Mitochondrial inverted repeats strongly correlate with lifespan: mtDNA inversions and aging. PLoS One, 8(9): e73318.
- Azpurua J, Yang JN, Van Meter M, Liu Z, Kim J, Lobo Ladd AA, Coppi AA, Gorbunova V, Seluanov A. 2013. IGF1R levels in the brain negatively correlate with longevity in 16 rodent species.Aging. 5(4): 304-14.
- Xiao T., Azpurua J., Hine C., Mao Z., Rempel M., Gorbunova V., and Seluanov A. 2013. High molecular weight hyaluronan mediates cancer resistance of the naked mole rat. Nature, 499(7458): 346-9.
- Azpurua J. and Seluanov A. 2013. Long-lived cancer-resistant rodents as new model species for cancer research. Frontiers in Genetics,3:319.
- VanMeter M., Seluanov A., and Gorbunova V. 2012. Forever young? Exploring the link between rapamycin, longevity and cancer. Cell Cycle,11(23): 4296-7.
- Gorbunova, V., Hine C., Tian X., Ablaeva J., Gudkov A.V., Nevo E., and Seluanov A. 2012. Cancer resistance in the blind mole rat is mediated by concerted necrotic cell death mechanism. Proc. Natl. Acad. Sci. USA, 109(47): 19392-6.
- Mao Z., Tian X., VanMeter M., Gorbunova V., and Seluanov A. 2012. Sirtuin 6 (SIRT6) rescues the decline of homologous recombination repair during replicative senescence.Proc. Natl. Acad. Sci. USA, 109(29): 11800-5.
- Anisimov VN, Bartke A, Barzilai N, Batin MA, Blagosklonny MV, Brown-Borg H, Budovskaya Y, Campisi J, Friguet B, Fraifeld V, Franceschi C, Gems D, Gladyshev V, Gorbunova V, Gudkov AV, Kennedy B, Konovalenko M, Kraemer B, Moskalev A, Petropoulos I, Pasyukova E, Rattan S, Rogina B, Seluanov A, Shaposhnikov M, Shmookler Reis R, Tavernarakis N, Vijg J, Yashin A, Zimniak P. 2012. The second international conference "genetics of aging and longevity" Aging. 4(5): 305-17.
- 2012. Replicatively senescent cells are arrested in G1 and G2 phases.Aging, 4(6): 431-5.
- 2012. SIRT6 rescues senescence-related decline in DNA repair by homologous recombination.Proc. Natl. Acad. Sci. USA, In press.
- 2012. Rad51 promoter targeted gene therapy is effective for in vivo visualization and treatment of cancer.Molecular therapy, 20(2):347-55.
- 2011. Adenoviral vector driven by a minimal Rad51 promoter is selective for p53-deficient tumor cells.PLOS One, 6(12):e28714.
- 2011. Repairing split ends: SIRT6, mono-ADP ribosylation and DNA repair.Aging, (9):829-35.
- 2011. Sirt6 overexpression induces massive apoptosis in cancer cells but not in normal cells.Cell Cycle, 10(18):3153-8.
- 2011. SIRT6 promotes DNA repair under stress by mono-ADP-ribosylating PARP1.Science, 17;332(6036):1443-6.*
*Comment in: Sirtuin 6 and DNA Repair, Guy Riddihough, Science Signaling 21 June 2011: ec177.
*Selected for Faculty of 1000 - 2010. Establishing primary adult fibroblast cultures from rodents.JoVE, 8;(43). pii: 2002. doi: 10.3791/2002.
- 2010. Analysis of DNA Double-Strand Break (DSB) Repair in Mammalian Cells.JoVE, Oct 5;(44). pii: 2033. doi: 10.3791/2033.
- 2009. Naked mole-rat cells are hypersensitive to contact inhibition – a clue to extraordinary cancer resistance.Proc. Natl. Acad. Sci, USA, 106(46):19352-7**
**Received Cozzarelli Prize for a paper of outstanding scientific excellence and originality published in 2009
**Selected for Faculty of 1000. - 2009. DNA repair by homologous recombination, but not by nonhomologous end joining, is elevated in breast cancer cells.Neoplasia, 11(7):683-691.
- 2009. Coevolution of telomerase activity and body mass in mammals: from mice to beavers.Mech. Aging. Dev., 130(1-2):3-9 (Invited review).
- 2008. Use of Rad51 promoter for targeted anticancer therapy.Proc. Natl. Acad. Sci. USA, 105(52) pp.20810-20815.
- 2008. Distinct tumor suppressor mechanisms evolve in rodent species that differ in size and lifespan.Aging Cell, 7(6) pp.813-823.
- 2008. DNA repair by nonhomologous end joining and homologous recombination during cell cycle in human cells.Cell Cycle, 7(18) pp.2902-2906.
- 2008. Comparison of nonhomologous end joining and homologous recombination in human cells. DNA Repair(Amst), 7(10) pp.1765-1771.
- 2008. Rodents for comparative aging studies: from mice to beavers.Age, 30(2-3) pp.111-119. (Invited review) 21.
- 2008. Genome-wide demethylation promotes triplet repeat instability independently of homologous recombination.DNA Repair (Amst), 7(2) pp.313-320.
- 2007. Changes in DNA repair during Aging.Nucleic Acids Res., 35(22) pp.7466-7474. (Invited review)
- 2007. TRF2 is required for repair of non-telomeric DNA double-strand breaks by homologous recombination,Proc. Natl. Acad. Sci. USA, 104(32) pp.13068-13073.
- 2007. Changes in the level and distribution of Ku proteins during cellular senescence.DNA Repair (Amst), 6(12) pp.1740-1748.
- 2007. Changes in the level and distribution of Ku proteins during cellular senescence.DNA Repair (Amst), 6(12) pp.1740-1748.
- 2007. Telomerase Activity Coevolves with Body Mass not Lifespan.,Aging Cell, 6(1) pp.45-52.
- 2007. Cell divisions are required for L1 retrotransposition.Mol. Cell. Biol., 27(4) pp.1264-1270.
- 2005. Making ends meet in old age: DSB repair and aging.,Mech. Aging. Dev., 126(6-7), pp.621-628. (Invited review).
- 2004. DNA repair by nonhomologous end joining becomes less efficient and more error-prone during cellular senescence.Proc. Natl. Acad. Sci. USA., 101(20), pp.7624-7629.
- 2004. Genome-wide demethylation destabilizes CTG/CAG trinucleotide repeats in mammalian cells.,Human Molecular Genetics, 13(23), pp.2979-2989. * Equal contribution.
- 2003. Telomerase as a growth promoting factor.Cell Cycle., 2(6), pp.534-537.
- 2003. Selectable system for monitoring the instability of CTG/CAG triplet repeats in mammalian cells.Mol. Cell Biol., 23(13) pp.4485-4493.
Book Chapters
- VanMeter M, Gorbunova V and Seluanov A. (2014) SIRT6: a promising target for cancer prevention and therapy, in “Anticancer Genes”, Editor: Grimm S., Springer.
- Gorbunova V. and Seluanov A. (2009) A comparison of senescence in mouse and human cells, in “Senescence and Cancer”, Editors: Sedivy J. and Adams P., Springer.
Patents
- Gorbunova V. Seluanov. A. (2018) Transposable Elements as Biomarkers of Aging and Disease. Ref. 1-19056. Pending.
- Gorbunova V. Seluanov. A. (2016) Compositions and Methods for Use in High Molecular Weight Hyaluronic Acid for Cancer Therapy. Ref. 1-11027-13043. Pending.
- Gorbunova V. Seluanov. A. (2016) Compositions and Methods for Recombinant Synthesis of High Molecular Weight Hyaluronic Acid. Patent number: 9315588, Date of Patent: April 19, 2016.
- Gorbunova V., Seluanov. A., and Hine C. (2008) Use of Rad51 promoter for transcriptionally targeted anticancer therapy. Invention disclosure # 6-1652, Pending.
