 |
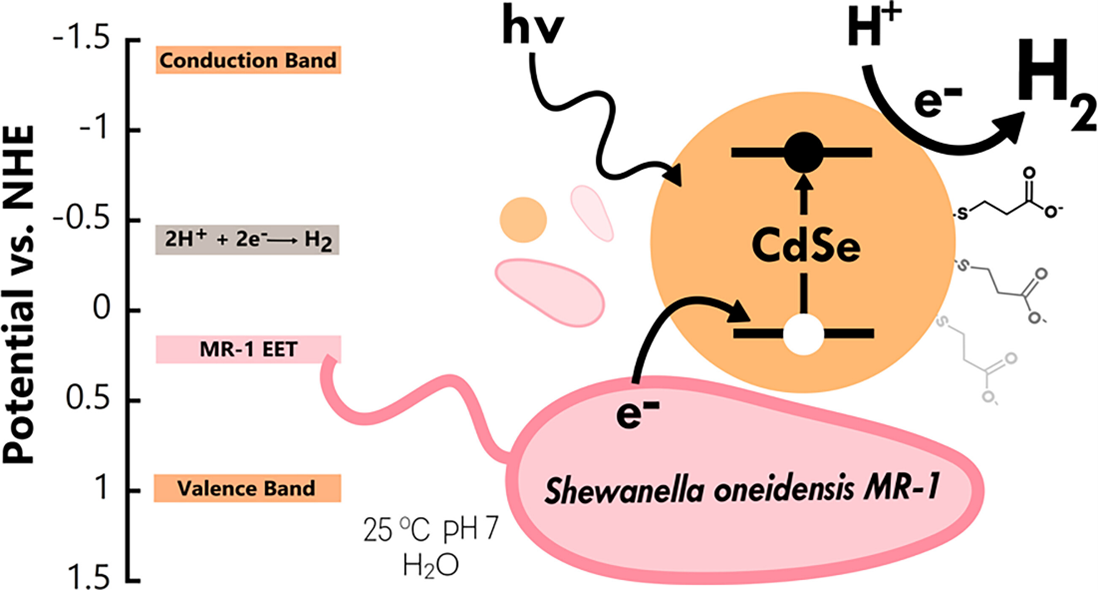
| In traditional three-component photochemical systems for hydrogen production, a molecular electron donor is employed in high excess in order to supply the system with electrons. In this project, we are avoiding this wasteful route to products by utilizing a living electron donor in the form of electrogenic bacteria that can provide a steady stream of metabolic electrons. In collaboration with the Krauss, Meyer and White labs, we are characterizing a novel bio-nano hybrid system that combines electrogenic bacteria and semiconductor nanoparticle catalysts for photochemical hydrogen production. Our contributions in the Bren lab include culturing bacteria and nanoparticles and monitoring hydrogen production under different conditions. We also employ different spectroscopic and imaging studies to better understand the interaction between the bacteria and nanoparticles in this exciting system. |
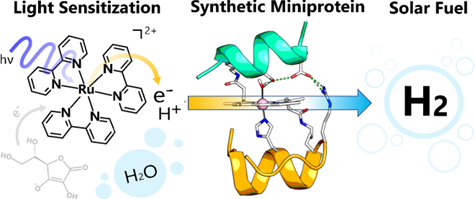 |
| Developing sustainable routes to alternative fuels is one of the major challenges facing humanity. Hydrogen is an energy-dense alternative fuel that is sustainable when synthesized from water in a light-driven reaction. The Bren group is working to characterize a series of biomolecular cobalt catalysts through electrochemical and photochemical methods. In our efforts to characterize and optimize these systems we explore the effects of buffer, pH, and component concentration on the activity of the system. In our ongoing work we are investigating new photosensitizers and catalysts for use in hydrogen evolution systems. In collaboration with the Krauss and Matson group, we work with developing photochemical systems that utilize CdSe quantum dot photocatalysts and polyoxovanadate clusters to enhance hydrogen production. Some current work in this area involves the investigation of in situ catalysts formed during photochemical reactions. |
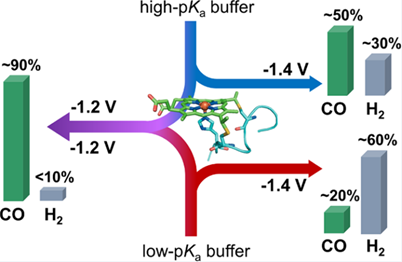 |
| The reduction of CO2 is a promising route to producing renewable fuels using an abundant carbon feedstock. Enzymes, known for catalyzing multi-electron, multi-proton reactions with high selectivity in water using earth-abundant metals, make attractive systems to emulate when developing reactions requiring controlled electron and proton delivery. We study the activity of the metalloporphyrin-containing biocatalyst cobalt microperoxidase-11 (CoMP11). CoMP11 is shown to significantly outperform other cobalt biomolecular catalysts for CO2 reduction, which may indicate that structure enhances activity in this system. We have also reported that the selectivity of the system can be tuned through buffer acidity and applied potential. Our main goal is to optimize activity and selectivity in photo- and electrochemical systems. Current work in the Bren lab is employing the effects of catalyst structure, buffer and applied potential to precisely control the selectivity of the complex CO2 reduction reaction. |
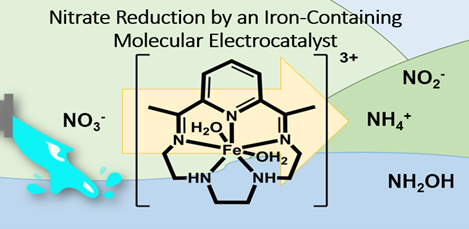 |
| Human contribution to excess environmental nitrogen species from use of fertilizers has led to an unhealthy level of nitrates and nitrites in our ground water. The downstream effects of these high NOy- levels contribute to algal blooms and hypoxic dead zones that decrease marine biodiversity. Nitrate and nitrite reduction are complicated multi- proton and electron reactions that requir a suitable and robust catalyst. In collaboration with the Matson lab, we study an iron complex FeN5H2 as a biomimetic electrocatalytic NOy- reducing catalyst in water. We are actively investigating the activity and mechanism of this impressive catalyst through different spectroscopic and electrochemical means. |
